Leucine
Branched Chain Amino Acids (Isoleucine, Leucine, Valine) are the three branched chain amino acids (BCAAs). Branched chain amino acids (BCAA) are essential amino acids and must be obtained from the diet (mainly meat, grains, and dairy).
Not only do the BCAAs account for almost 50% of muscle protein, but they have many metabolic functions.
BCAAs act as substrates for protein synthesis, energy production, neurotransmitter production, glucose metabolism, and the immune response. They are also involved in stimulation of albumin and glycogen synthesis, improvement of insulin resistance, inhibition of free radical production, and hepatocyte apoptosis with liver regeneration.
Unlike most amino acids, the initial step of BCAA metabolism does not take place in the liver. After dietary intake, BCAAs remain in circulation and are taken up by skeletal muscle, the heart, kidney, diaphragm, and adipose tissue for immediate metabolism. BCAAs are transaminated into α-keto acids and used within the tissues or released into circulation. The liver and other organs can then further catabolize these α-keto acids. The complete oxidation of valine yields succinyl CoA, and leucine and isoleucine produce acetyl CoA for use in the citric acid cycle. Isoleucine also produces propionyl CoA and succinyl CoA.
Skeletal muscle is a major site of BCAA utilization. During exercise, catabolism of the BCAAs is elevated; β-aminoisobutyric acid (β-AIB) is a metabolite of valine released during exercise. There is much published literature on the use of BCAAs for muscle protein synthesis, however it’s been shown that BCAA supplementation alone does not enhance muscle protein synthesis better than the consumption of a complete, high quality food protein containing the full spectrum of essential amino acids.
Of the three BCAA, leucine may have the most immediate impact. Leucine is one of the few amino acids that is completely oxidized in the muscle for energy, generating more ATP molecules than glucose. Additionally, leucine can be used to synthesize fatty acids in adipose tissue, and generates HMG CoA, an intermediate in the synthesis of cholesterol.
Leucine also stimulates insulin secretion and promotes protein synthesis in the liver, muscle, and skin. BCAAs, grouped in patterns and as single biomarkers, have been studied as predictors of obesity, insulin resistance, type 2 diabetes, metabolic syndrome, and cardiovascular disease outcomes.
References:
- Gorska-Warsewicz H, Laskowski W, Kulykovets O, Kudlinska Chylak A, Czeczotko M, Rejman K. Food Products as Sources of Protein and Amino Acids-The Case of Poland. Nutrients. 2018;10(12):1977.
- Santos CS, Nascimento FEL. Isolated branched-chain amino acid intake and muscle protein synthesis in humans: a biochemical review. Einstein. 2019;17(3):eRB4898.
- Grüngreiff K. Branched amino acids and zinc in the nutrition of liver cirrhosis. J Clin Exp Hepatol. 2018;8(4):480.
- Holecek M. Branched-chain amino acids in health and disease: metabolism, alterations in blood plasma, and as supplements. Nutr Metab. 2018;15(1):33.
- Gropper S SJ, Groff J. Adv Nutr Human Metab. 5th ed. Belmont, CA: Wadsworth, Cengage Learning; 2009.
- Roberts LD, Bostrom P, O'Sullivan JF, et al. betaAminoisobutyric acid induces browning of white fat and hepatic beta-oxidation and is inversely correlated with cardiometabolic risk factors. Cell Metab. 2014;19(1):96-108.
- Mero A. Leucine supplementation and intensive training. Sports Med. 1999;27(6):347-358.
- Nie C, He T, Zhang W, Zhang G, Ma X. Branched Chain Amino Acids: Beyond Nutrition Metabolism. Int J Mol Sci. 2018;19(4).
- Ntzouvani A, Nomikos T, Panagiotakos D, et al. Amino acid profile and metabolic syndrome in a male Mediterranean population: A cross-sectional study. Nutr Metab Cardiovasc Dis. 2017;27(11):1021-1030.
- Yennawar N, Dunbar J, Conway M, Hutson S, Farber G. The structure of human mitochondrial branched-chain aminotransferase. Biol Crystallog. 2001;57(Pt 4):506-515.
- Duran M, Wadman SK. Thiamine-responsive inborn errors of metabolism. J Inher Metab Dis. 1985;8 Suppl 1:70-75.
- Chuang DT, Ku LS, Cox RP. Thiamin-responsive maplesyrup-urine disease: decreased affinity of the mutant branched-chain alpha-keto acid dehydrogenase for alphaketoisovalerate and thiamin pyrophosphate. Proc Nat Acad Sci USA. 1982;79(10):3300-3304.
- Fernhoff PM, Lubitz D, Danner DJ, et al. Thiamine response in maple syrup urine disease. Ped Res. 1985;19(10):1011-1016.
- Solmonson A, DeBerardinis RJ. Lipoic acid metabolism and mitochondrial redox regulation. J Biol Chem. 2018;293(20):7522-7530.
- Jurgens P, Schwartau M, Doehn M. [Disorders of amino acid metabolism in a patient with identified thiamine deficiency]. Infusionstherapie klinische Ernahrung. 1982;9(6):312-316.
- Brunetti-Pierri N, Lanpher B, Erez A, et al. Phenylbutyrate therapy for maple syrup urine disease. Human Mol Gen. 2011;20(4):631-640.
- Girish BN, Rajesh G, Vaidyanathan K, Balakrishnan V. Alterations in plasma amino acid levels in chronic pancreatitis. JOP : J Pancreas. 2011;12(1):11-18.
- Shaw S, Lieber CS. Plasma amino acids in the alcoholic: nutritional aspects. Alc Clin Exp Reds. 1983;7(1):22-27.
- Kawaguchi T, Nagao Y, Abe K, et al. Effects of branched-chain amino acids and zinc-enriched nutrients on prognosticators in HCV-infected patients: a multicenter randomized controlled trial. Molec Med Rep. 2015;11(3):2159-2166.
- Park YK, Linkswiler H. Effect of vitamin B6 depletion in adult man on the plasma concentration and the urinary excretion of free amino acids. JNutr. 1971;101(2):185-191.
- Vannucchi H, Moreno FS, Amarante AR, de Oliveira JE, Marchini JS. Plasma amino acid patterns in alcoholic pellagra patients. Alcohol Alcoholism. 1991;26(4):431-436.
Lab Results Explained and Tracked
What does it mean if your Leucine result is too high?
High protein intake may elevate BCAAs. In the catabolism of BCAAs, branched chain aminotransferase and branched chain alpha ketoacid dehydrogenase complex (BCKDC) require several cofactors such as vitamin B6, vitamin B1, and lipoic acid. Therefore, functional need for these cofactors may contribute to high levels of BCAAs.
BCAAs can be elevated due to a rare inborn error of metabolism. Maple Syrup Urine Disease is an inherited disorder of branched chain amino acid metabolism due to deficiency of the BCKDC complex.
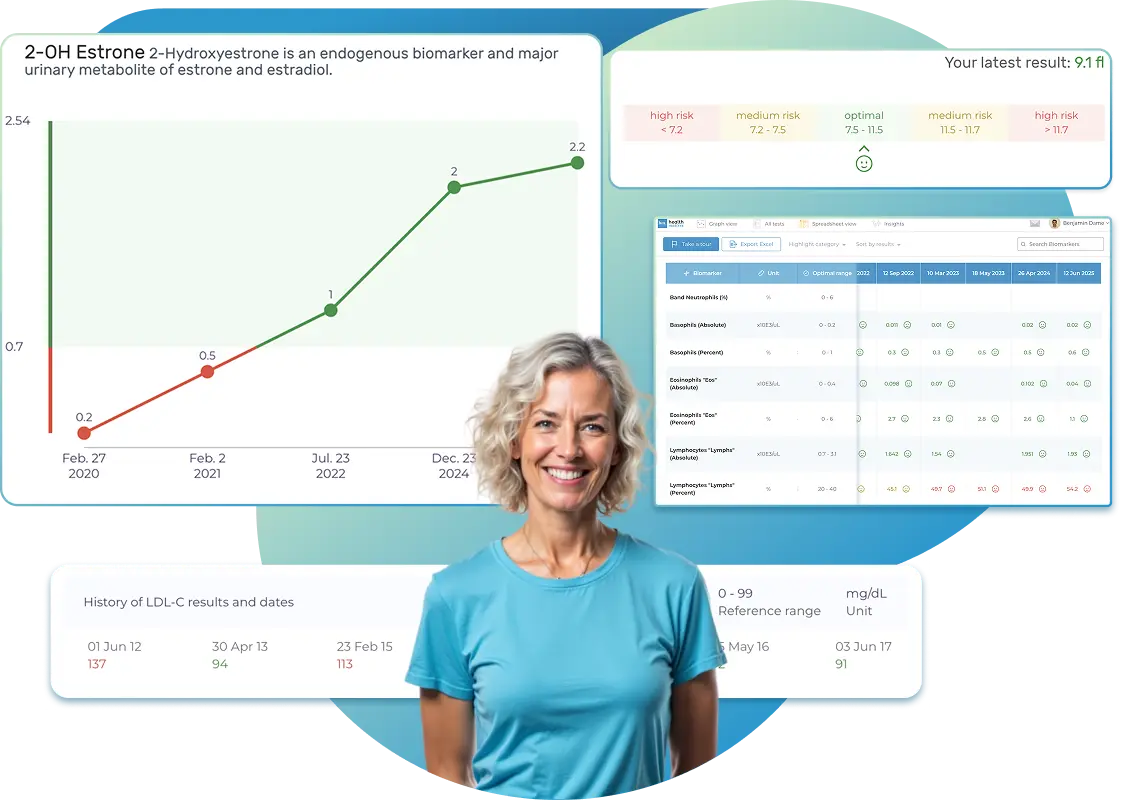
All Your Lab Results.
One Simple Dashboard.
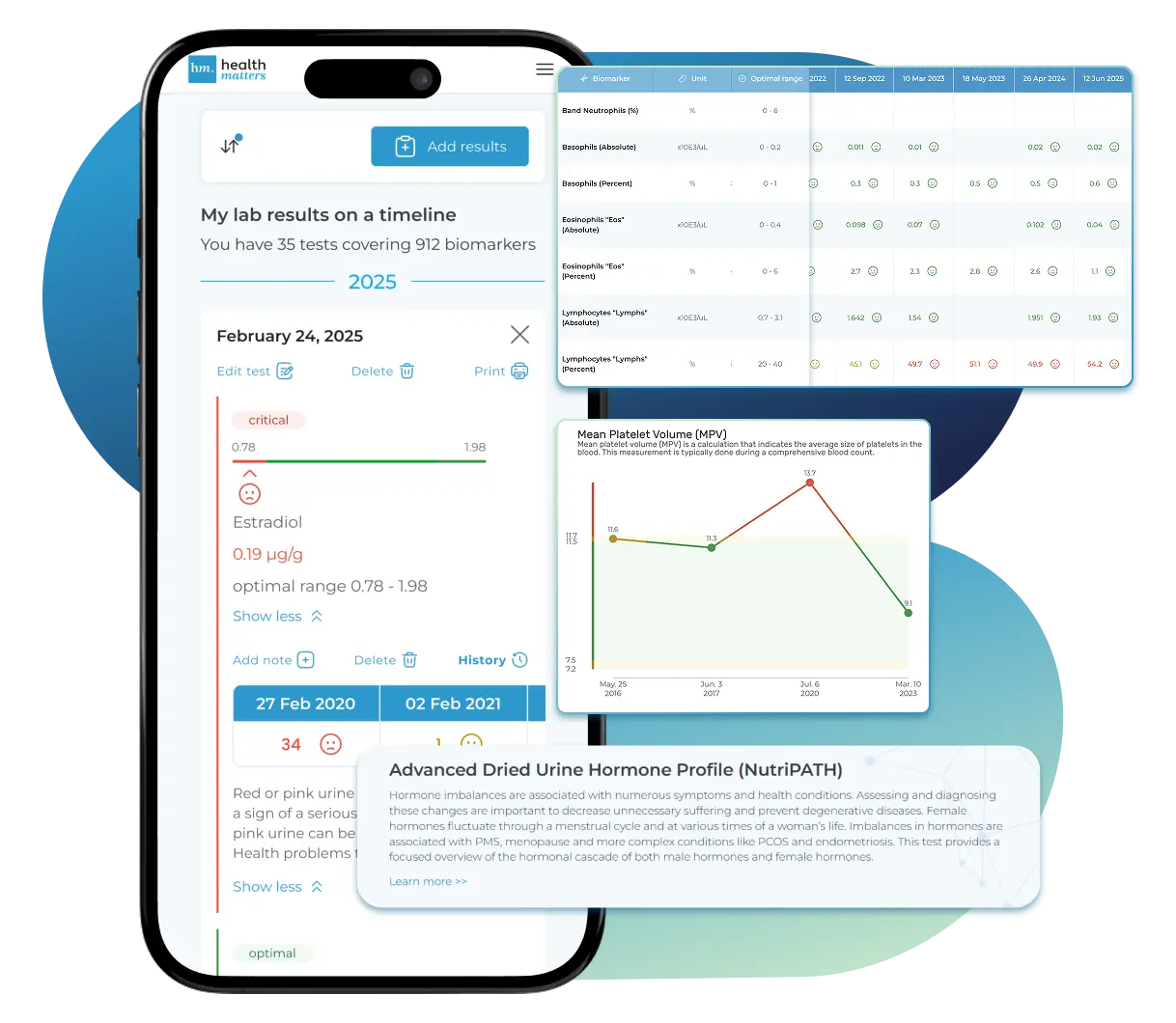
Import, Track, and Share Your Lab Results Easily
Import, Track, and Share Your Lab Results
Import lab results from multiple providers, track changes over time, customize your reference ranges, and get clear explanations for each result. Everything is stored securely, exportable in one organized file, and shareable with your doctor—or anyone you choose.
Cancel or upgrade anytime
What does it mean if your Leucine result is too low?
Low levels of essential BCAA may indicate a poor-quality diet, or maldigestion due to deficient digestive peptidase activity or pancreatic dysfunction.
Low levels of leucine can be seen after significant aerobic exercise or strength training.
Supplementation with zinc, vitamin B3, and vitamin B6, has improved outcomes in various conditions where low levels of BCAA’s have been associated.
Article Review & Sources
All our content is backed by peer-reviewed studies, academic research, and trusted medical sources. We're committed to accuracy and transparency — see our editorial policy for details.
Laboratories
Bring All Your Lab Results Together — In One Place
We accept reports from any lab, so you can easily collect and organize all your health information in one secure spot.
Pricing Table
Gather Your Lab History — and Finally Make Sense of It
Finally, Your Lab Results Organized and Clear
Personal plans
$79/ year
Advanced Plan
Access your lab reports, explanations, and tracking tools.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 5 reports entered for you
- Cancel or upgrade anytime
$250/ once
Unlimited Account
Pay once, access everything—no monthly fees, no limits.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 10 reports entered for you
- No subscriptions. No extra fees.
$45/ month
Pro Monthly
Designed for professionals managing their clients' lab reports
- Import lab results from any provider
- Track lab results for multiple clients
- Customize reference ranges per client
- Export lab histories and reports
- Begin with first report entered by us
- Cancel or upgrade anytime
About membership
What's included in a Healthmatters membership
 Import Lab Results from Any Source
Import Lab Results from Any Source
 See Your Health Timeline
See Your Health Timeline
 Understand What Your Results Mean
Understand What Your Results Mean
 Visualize Your Results
Visualize Your Results
 Data Entry Service for Your Reports
Data Entry Service for Your Reports
 Securely Share With Anyone You Trust
Securely Share With Anyone You Trust
Let Your Lab Results Tell the Full Story
Once your results are in one place, see the bigger picture — track trends over time, compare data side by side, export your full history, and share securely with anyone you trust.





Bring all your results together to compare, track progress, export your history, and share securely.
What Healthmatters Members Are Saying
We implement proven measures to keep your data safe.
At HealthMatters, we're committed to maintaining the security and confidentiality of your personal information. We've put industry-leading security standards in place to help protect against the loss, misuse, or alteration of the information under our control. We use procedural, physical, and electronic security methods designed to prevent unauthorized people from getting access to this information. Our internal code of conduct adds additional privacy protection. All data is backed up multiple times a day and encrypted using SSL certificates. See our Privacy Policy for more details.

















