Anti-Glucose regulated protein 78 (IgG + IgA)
What is Glucose-regulated protein 78 (GRP78)?
Glucose-regulated protein 78 (GRP78) is a stress-inducible molecular chaperone expressed within the endoplasmic reticulum ("ER) where it acts as a master regulator of the unfolded protein response ("UPR") pathway.
The 78 kDa glucose-regulated protein (GRP78) is an endoplasmic reticulum (ER)-resident molecular chaperone. GRP78 is a member of the 70 kDa heat shock family of proteins involved in correcting and clearing misfolded proteins in the ER. In response to cellular stress, GRP78 escapes from the ER and moves to the plasma membrane where it (a) functions as a receptor for many ligands, and (b) behaves as an autoantigen for autoantibodies that contribute to human disease and cancer.
→ Chaperones are proteins that guide proteins along the proper pathways for folding.
→ Endoplasmic reticulum is a network of membranes inside a cell through which proteins and other molecules move.
→ The unfolded protein response is the cells’ way of maintaining the balance of protein folding in the endoplasmic reticulum, which is the section of the cell designated for folding proteins with specific destinations such as other organelles or to be secreted by the cell.
At times of ER (= endoplasmic reticulum) stress, activation of the unfolded protein response, a multimolecular pathway, limits proteotoxicity (= a pathology that develops due to damaged or misfolded protein accumulations).
In malignancies, including multiple myeloma which is characterized by an accumulation of misfolded immunoglobulins, GRP78 expression is increased, with notable translocation of GRP78 to the cell surface. Studies suggest cell-surface GRP78 (csGRP78) to be of prognostic significance with emerging evidence that it interacts with a myriad of co-ligands to activate signaling pathways promoting cell proliferation and survival or apoptosis.
→ Cell surface GRP78 is preferably expressed in cancer cells and stressed endothelial cells
References:
Lemke MR, Glatzel M, Henneberg AE. Antimicroglia antibodies in sera of Alzheimer's disease patients. Biol Psychiatry. 1999 Feb 15;45(4):508-11. doi: 10.1016/s0006-3223(97)00456-3. PMID: 10071725.
Ninkovic S, Harrison SJ, Quach H. Glucose-regulated protein 78 (GRP78) as a potential novel biomarker and therapeutic target in multiple myeloma. Expert Rev Hematol. 2020 Nov;13(11):1201-1210. doi: 10.1080/17474086.2020.1830372. Epub 2020 Oct 18. PMID: 32990063.
Slavisa Ninkovic, Simon J. Harrison & Hang Quach (2020) Glucose-regulated protein 78 (GRP78) as a potential novel biomarker and therapeutic target in multiple myeloma, Expert Review of Hematology, 13:11, 1201-1210, DOI: 10.1080/17474086.2020.1830372
Xia, S., Duan, W., Liu, W. et al. GRP78 in lung cancer. J Transl Med 19, 118 (2021). https://doi.org/10.1186/s12967-021-02786-6
Gonzalez-Gronow, M, Gopal, U, Austin, RC, Pizzo, SV. Glucose-regulated protein (GRP78) is an important cell surface receptor for viral invasion, cancers, and neurological disorders. IUBMB Life. 2021; 73: 843– 854. https://doi.org/10.1002/iub.2502
Read A, Schröder M. The Unfolded Protein Response: An Overview. Biology (Basel). 2021 Apr 29;10(5):384. doi: 10.3390/biology10050384. PMID: 33946669; PMCID: PMC8146082.
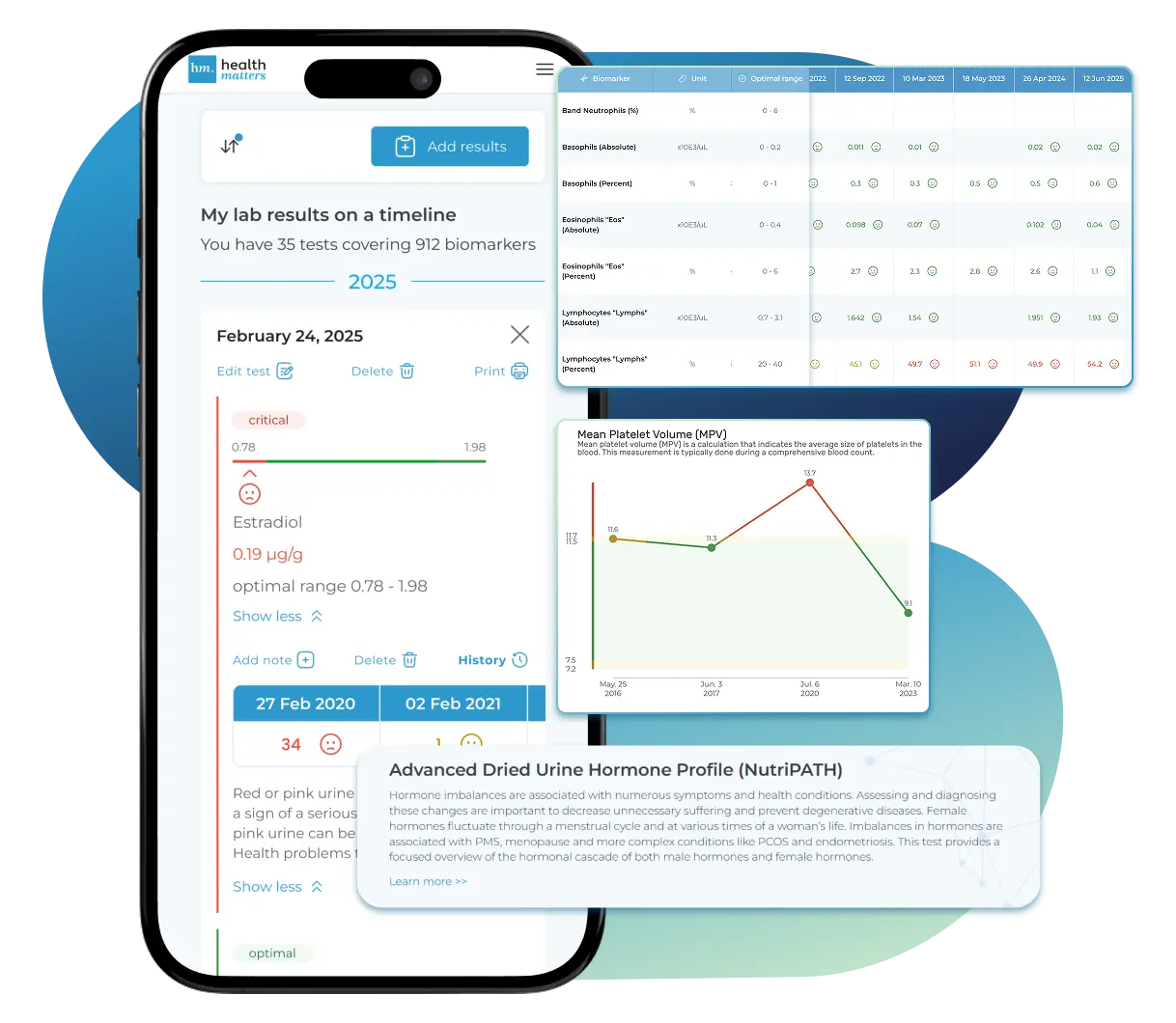
Lab Results Explained and Tracked
What does it mean if your Anti-Glucose regulated protein 78 (IgG + IgA) result is too high?
Studies show that antibodies targeting glucose-regulated protein 78 are able to activate brain microvascular endothelial cells and induce protein extravasation in cell lines and in mice with neuromyelitis optica. Thus, these findings suggest that glucose-regulated protein 78–targeted antibodies could instigate blood brain barrier breakdown and development of hallmark anti–aquaporin-4 autoantibody pathology. Therefore, the application of these antibodies may be useful to disrupt the blood brain barrier for transit of treatments for many CNS diseases.
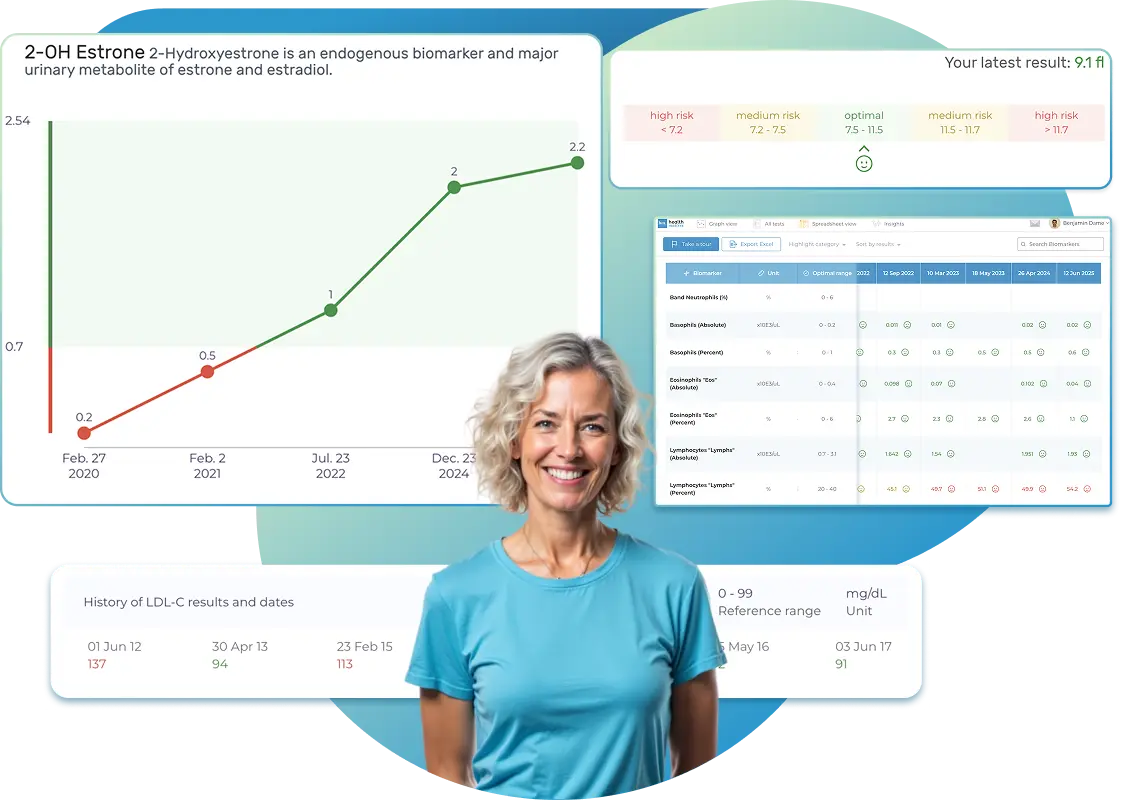
All Your Lab Results.
One Simple Dashboard.
Import, Track, and Share Your Lab Results Easily
Import, Track, and Share Your Lab Results
Import lab results from multiple providers, track changes over time, customize your reference ranges, and get clear explanations for each result. Everything is stored securely, exportable in one organized file, and shareable with your doctor—or anyone you choose.
Cancel or upgrade anytime
Article Review & Sources
All our content is backed by peer-reviewed studies, academic research, and trusted medical sources. We're committed to accuracy and transparency — see our editorial policy for details.
Laboratories
Bring All Your Lab Results Together — In One Place
We accept reports from any lab, so you can easily collect and organize all your health information in one secure spot.
Pricing Table
Gather Your Lab History — and Finally Make Sense of It
Finally, Your Lab Results Organized and Clear
Personal plans
$79/ year
Advanced Plan
Access your lab reports, explanations, and tracking tools.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 5 reports entered for you
- Cancel or upgrade anytime
$250/ once
Unlimited Account
Pay once, access everything—no monthly fees, no limits.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 10 reports entered for you
- No subscriptions. No extra fees.
$45/ month
Pro Monthly
Designed for professionals managing their clients' lab reports
- Import lab results from any provider
- Track lab results for multiple clients
- Customize reference ranges per client
- Export lab histories and reports
- Begin with first report entered by us
- Cancel or upgrade anytime
About membership
What's included in a Healthmatters membership
 Import Lab Results from Any Source
Import Lab Results from Any Source
 See Your Health Timeline
See Your Health Timeline
 Understand What Your Results Mean
Understand What Your Results Mean
 Visualize Your Results
Visualize Your Results
 Data Entry Service for Your Reports
Data Entry Service for Your Reports
 Securely Share With Anyone You Trust
Securely Share With Anyone You Trust
Let Your Lab Results Tell the Full Story
Once your results are in one place, see the bigger picture — track trends over time, compare data side by side, export your full history, and share securely with anyone you trust.





Bring all your results together to compare, track progress, export your history, and share securely.
What Healthmatters Members Are Saying
We implement proven measures to keep your data safe.
At HealthMatters, we're committed to maintaining the security and confidentiality of your personal information. We've put industry-leading security standards in place to help protect against the loss, misuse, or alteration of the information under our control. We use procedural, physical, and electronic security methods designed to prevent unauthorized people from getting access to this information. Our internal code of conduct adds additional privacy protection. All data is backed up multiple times a day and encrypted using SSL certificates. See our Privacy Policy for more details.

















