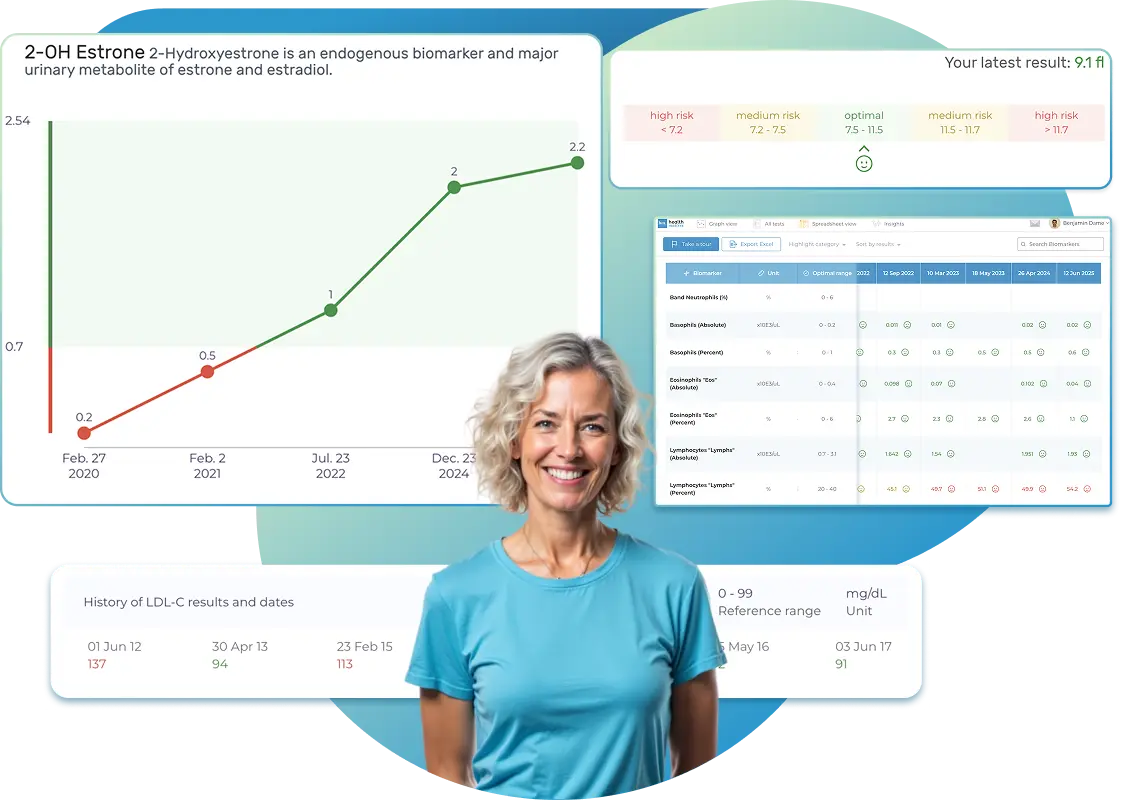
Research and clinical studies show that the 2-hydroxylated estrogens (2-OH E2 and 2-OH E1) are a safer pathway of hydroxylation than the 4-hydroxyestrogens (4-OH E2 and 4-OH E1), which bind to and damage DNA, leading to mutations that are associated with increased breast cancer risk. If either 4-OH-E2 or 4-OH-E1 are higher than reference range, and not well methylated, this may indicate higher risk for DNA damage, mutations, and risk for developing breast cancer.
Hydroxylation of estrogens in the 2-position of estradiol (2-OH-E2) and estrone (2-OH-E1) is considered a safer pathway for metabolism than the 4-position hydroxylations of these estrogens. The 2-position hydroxylations (considered safer) are increased with cruciferous vegetables (and extracts of them) as well as iodine. The most commonly used are indole-3-carbinol (I3C) and its metabolite diindolylmethane (DIM). Iodine also increases the 2-hydroxylation of estrogens, with a slight increase in 4-hydroxylation. The more dangerous 4-hydroxylated estrogen metabolism is enhanced by exposure to environmental toxins, mostly petrochemical-based products but also heavy metals, that induce 4-hydroxylation pathway enzymes, and cause formation of Reactive Oxygen Species (ROS) that co-oxidize the catechol estrogens to more dangerous estrogen quinones.
Lab Results Explained and Tracked
What does it mean if your 2-OH Estradiol result is too high?
Research and clinical studies show that the 2-hydroxylated estrogens (2-OH E2 and 2-OH E1) are a safer pathway of hydroxylation than the 4- hydroxyestrogens (4-OH E2 and 4-OH E1), which are considered more toxic as they bind to DNA causing mutations that are associated with increased breast cancer risk.
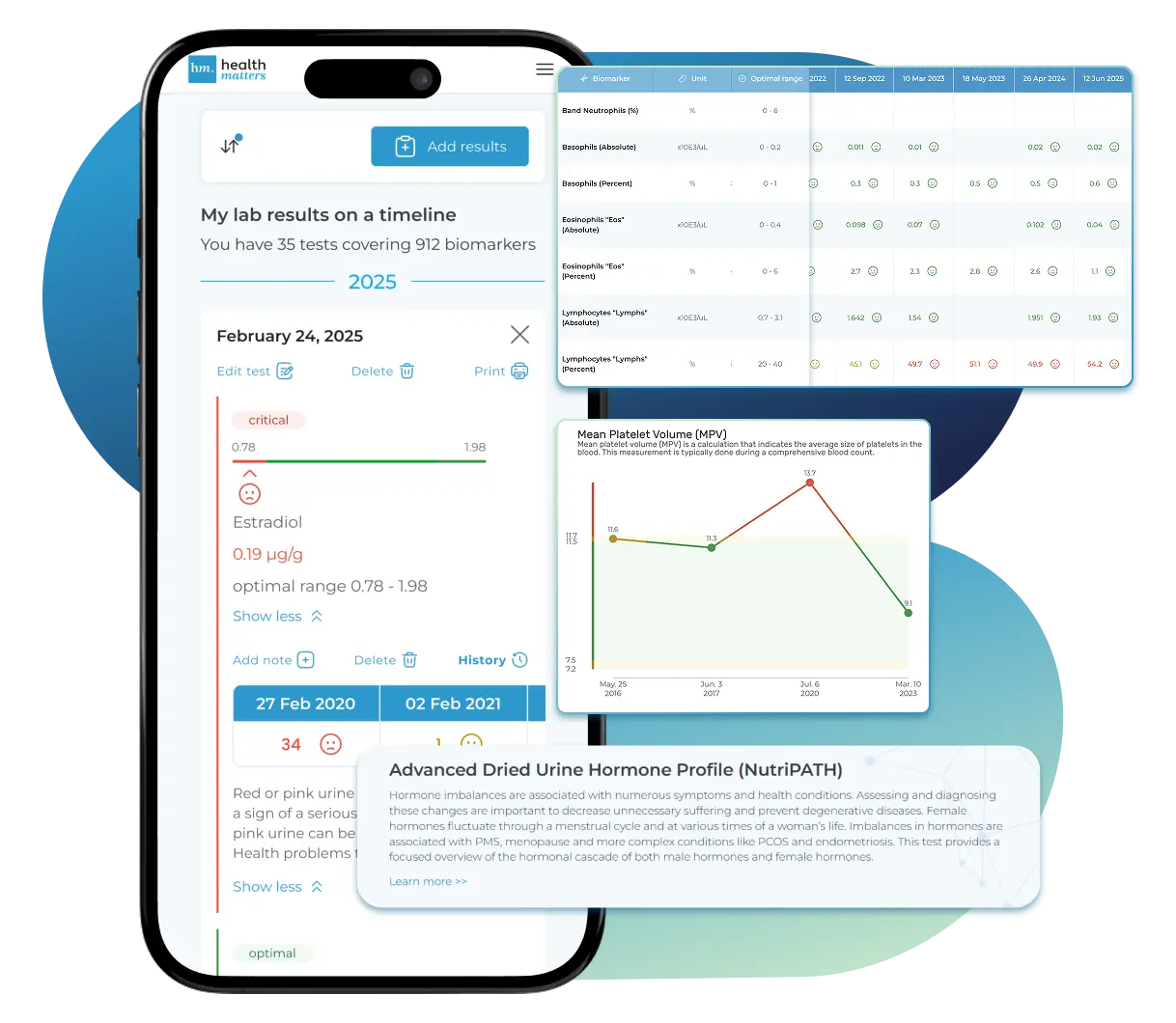
All Your Lab Results.
One Simple Dashboard.
Import, Track, and Share Your Lab Results Easily
Import, Track, and Share Your Lab Results
Import lab results from multiple providers, track changes over time, customize your reference ranges, and get clear explanations for each result. Everything is stored securely, exportable in one organized file, and shareable with your doctor—or anyone you choose.
Cancel or upgrade anytime
Article Review & Sources
All our content is backed by peer-reviewed studies, academic research, and trusted medical sources. We're committed to accuracy and transparency — see our editorial policy for details.
Laboratories
Bring All Your Lab Results Together — In One Place
We accept reports from any lab, so you can easily collect and organize all your health information in one secure spot.
Pricing Table
Gather Your Lab History — and Finally Make Sense of It
Finally, Your Lab Results Organized and Clear
Personal plans
$79/ year
Advanced Plan
Access your lab reports, explanations, and tracking tools.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 5 reports entered for you
- Cancel or upgrade anytime
$250/ once
Unlimited Account
Pay once, access everything—no monthly fees, no limits.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 10 reports entered for you
- No subscriptions. No extra fees.
$45/ month
Pro Monthly
Designed for professionals managing their clients' lab reports
- Import lab results from any provider
- Track lab results for multiple clients
- Customize reference ranges per client
- Export lab histories and reports
- Begin with first report entered by us
- Cancel or upgrade anytime
About membership
What's included in a Healthmatters membership
 Import Lab Results from Any Source
Import Lab Results from Any Source
 See Your Health Timeline
See Your Health Timeline
 Understand What Your Results Mean
Understand What Your Results Mean
 Visualize Your Results
Visualize Your Results
 Data Entry Service for Your Reports
Data Entry Service for Your Reports
 Securely Share With Anyone You Trust
Securely Share With Anyone You Trust
Let Your Lab Results Tell the Full Story
Once your results are in one place, see the bigger picture — track trends over time, compare data side by side, export your full history, and share securely with anyone you trust.





Bring all your results together to compare, track progress, export your history, and share securely.
What Healthmatters Members Are Saying
We implement proven measures to keep your data safe.
At HealthMatters, we're committed to maintaining the security and confidentiality of your personal information. We've put industry-leading security standards in place to help protect against the loss, misuse, or alteration of the information under our control. We use procedural, physical, and electronic security methods designed to prevent unauthorized people from getting access to this information. Our internal code of conduct adds additional privacy protection. All data is backed up multiple times a day and encrypted using SSL certificates. See our Privacy Policy for more details.

















