What is Boron?
Boron is a naturally occurring trace mineral found in soil, food, and water. Although not officially classified as an “essential” nutrient in humans by major regulatory bodies, mounting research suggests that boron plays a significant role in maintaining optimal health. It influences several biological functions, particularly those related to bone health, hormone activity, brain function, and the body’s use of other minerals such as calcium, magnesium, and phosphorus.
In urine testing, boron levels reflect recent intake or exposure. The "Essential Elements; urine" panel by Mosaic Diagnostics assesses urinary excretion of boron and other minerals to help determine whether the body is absorbing and processing these nutrients appropriately, or whether there is evidence of excessive intake, deficiency, or imbalance.
Why Boron Matters for Health
1. Bone and Joint Health
Boron appears to assist in the metabolism of calcium, magnesium, and vitamin D—key nutrients for building and maintaining strong bones. It has been studied for its potential role in reducing the risk of osteoporosis and improving bone mineral density, particularly in postmenopausal women. Boron may also support joint health by influencing inflammatory pathways and the metabolism of sex hormones.
2. Hormonal Balance
Boron is involved in the regulation of steroid hormones, including estrogen and testosterone. Some studies have shown that adequate boron intake may help increase free testosterone levels in men and support estrogen balance in women. This makes it of interest in hormone optimization and menopausal support.
3. Brain and Cognitive Function
Boron may affect brain function, particularly areas involving attention, short-term memory, and motor control. Low boron status has been associated with poorer performance in cognitive tasks, while higher intakes are linked with improved mental alertness.
4. Inflammation and Immune Support
Boron may modulate inflammatory responses and influence the activity of enzymes such as cyclooxygenase (COX), which are involved in the production of inflammatory mediators. It may also support immune function and wound healing.
5. Nutrient Utilization
Boron supports the body’s use of other minerals. It may help regulate the transport and effectiveness of calcium, magnesium, and phosphorus—nutrients central to muscle function, nerve conduction, and metabolic health.
How Boron Is Measured in Urine
Urinary boron testing reflects short-term intake or exposure, often over the past 24–48 hours. It can be affected by:
-
Dietary intake (e.g., fruits, nuts, legumes, wine, and leafy greens are boron-rich)
-
Supplement use (boron is present in many multivitamins or bone-support formulas)
-
Environmental exposures (e.g., boron compounds used in certain industrial processes or cleaning products)
The urine test by Mosaic Diagnostics provides a quantitative measure of how much boron is being excreted in urine. This helps clinicians evaluate whether an individual’s intake is adequate, deficient, or potentially excessive.
Interpreting Your Boron Result
Elevated Urinary Boron
-
May indicate high dietary or supplemental intake
-
Could be a result of recent exposure to boron-containing products (industrial, environmental, or cosmetic)
-
Rarely, very high levels may be associated with toxicity, which might include symptoms like nausea, headache, skin irritation, or hormonal disruptions—though toxicity from dietary sources is uncommon
-
In many cases, a slightly elevated result in an otherwise healthy person is not considered harmful, particularly if supplementation was recent
Low or Decreased Urinary Boron
-
May reflect inadequate dietary intake
-
Could indicate poor absorption or increased utilization by the body
-
Associated with suboptimal bone mineral metabolism and reduced effectiveness of vitamin D and magnesium
-
May contribute to hormonal imbalances, decreased cognitive performance, or increased risk of inflammatory conditions
-
May warrant dietary adjustments or low-dose supplementation if clinically appropriate
What Affects Boron Levels?
Several factors can influence urinary boron levels:
-
Diet: Fruits (especially apples, grapes, pears), avocados, nuts, legumes, and leafy greens are rich in boron
-
Supplementation: Even small doses of boron in supplements can raise urinary levels
-
Hydration status: Affects urine concentration, which is why creatinine correction may be used
-
Renal function: Boron is excreted via the kidneys, so impaired kidney function can alter excretion rates
-
Environmental exposure: Contact with boron-based industrial products or detergents
Summary
Boron is a biologically active trace mineral that plays a subtle yet important role in supporting bone integrity, hormone regulation, cognitive function, and inflammation control. The "Essential Elements; urine" test by Mosaic Diagnostics provides insight into your body’s recent boron exposure and excretion.
A result within the reference range generally reflects healthy intake and metabolism. Elevated or decreased levels should be interpreted in the context of symptoms, diet, supplement use, and other mineral levels. If your result is abnormal, your healthcare provider may recommend dietary changes, targeted supplementation, or further testing to assess nutrient balance and overall metabolic health.
Let me know if you’d like this adapted for clinicians, or if you want companion descriptions for other elements in the panel (like magnesium, zinc, or lithium).
Lab Results Explained and Tracked
What does it mean if your Boron result is too high?
Boron (B) is introduced to the body mainly through food (noncitrus fruits, leafy vegetables, nuts, legumes, wine, cider, beer) and drinking water but is also found in food preservatives (sodium borate), and insecticides (boric acid). Evidence for biological essentiality in animals (including humans) has been presented. It has been proposed that boron contributes to living systems by acting indirectly as a proton donor and that it exerts a particular influence on cell membrane and structure and function. In humans boron is responsible for the hydroxylation of various substances in the body. It may enhance the production of various hormones such as testosterone, estrogen, DHEA, and 1,25 dihydroxycholecalciferol. Boron is very readily absorbed and excreted in the urine yet its concentration remains quite low in the serum. Based on urinary recovery findings, more than 90% of ingested B is usually absorbed. Boron is distributed throughout the tissues and organs of animals and humans at concentrations mostly between 4.6 and 55.5 nmol (0.05 and 0.6 μg)/g fresh weight. Among the organs that contain the highest amounts of B are bone, spleen, and thyroid. It appears to be most concentrated in the thyroid gland.
Boron has a low order of toxicity even with intakes as high as 40mg/day in some parts of the world. However, high body burden of the element may be harmful, especially to young animals (including human neonates). Reports have shown that when doses equivalent to more than 46 mmol (0.5 g) B daily were consumed, disturbances in appetite, digestion, and health occurred. Acute toxicity signs include nausea, vomiting, diarrhea, dermatitis, and lethargy. High B ingestion also induces riboflavinuria.
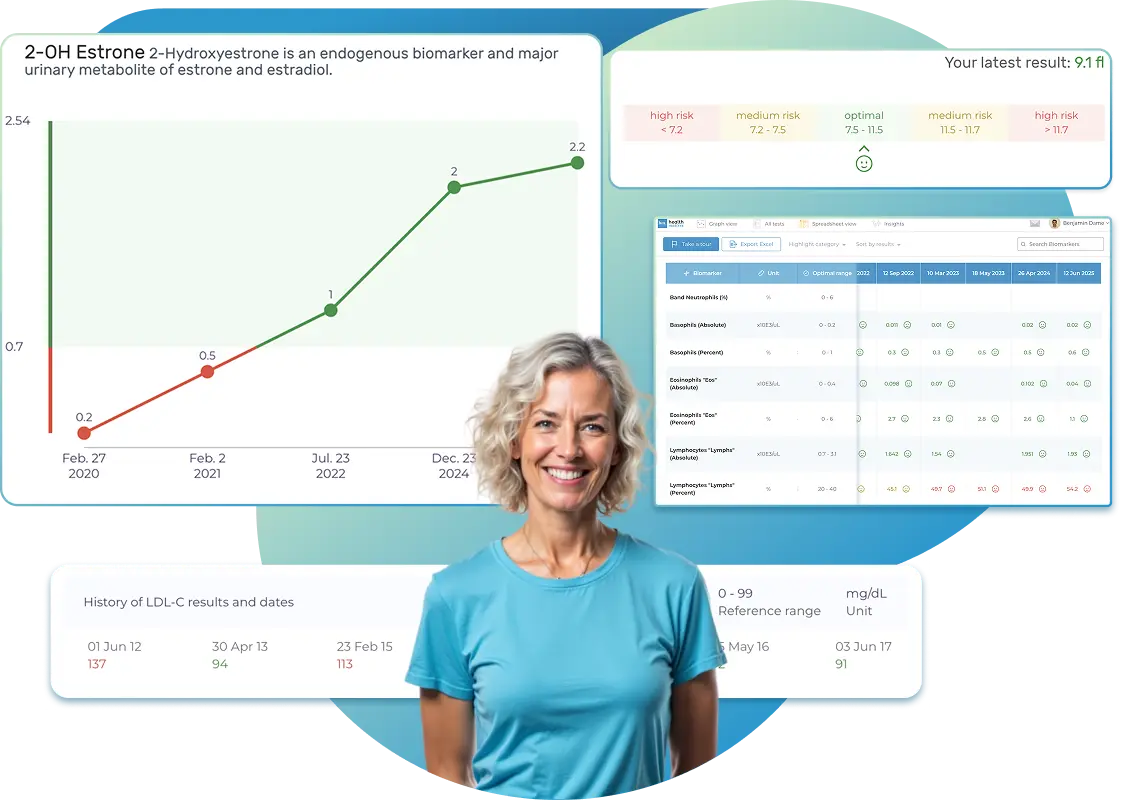
All Your Lab Results.
One Simple Dashboard.
Import, Track, and Share Your Lab Results Easily
Import, Track, and Share Your Lab Results
Import lab results from multiple providers, track changes over time, customize your reference ranges, and get clear explanations for each result. Everything is stored securely, exportable in one organized file, and shareable with your doctor—or anyone you choose.
Cancel or upgrade anytime
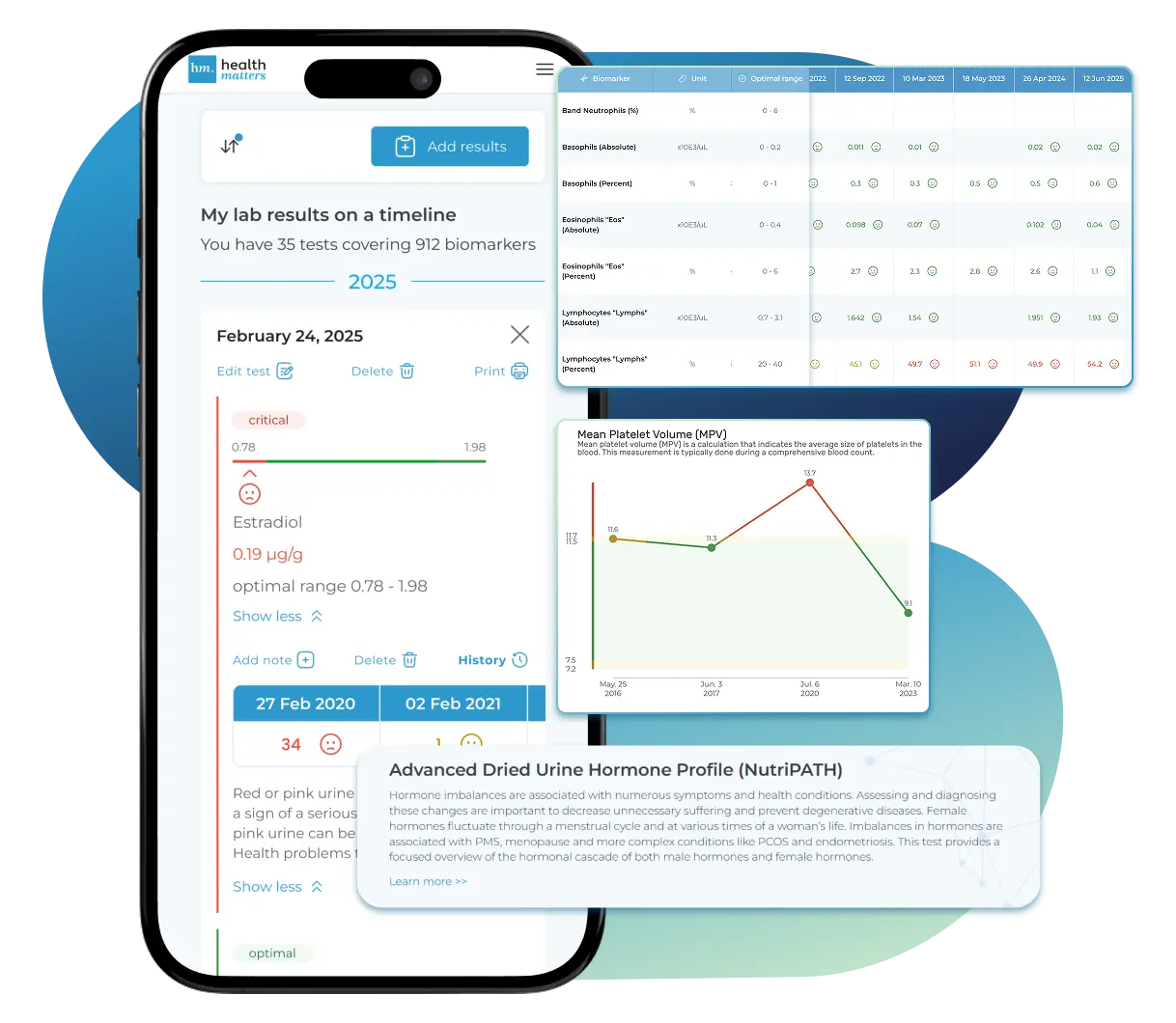
What does it mean if your Boron result is too low?
What does a low boron level mean?
A low urinary boron level suggests that your body may be getting insufficient boron from diet, supplements, or other environmental sources, or that it is not effectively absorbing or utilizing this trace mineral. Since boron is excreted primarily through urine, decreased levels typically reflect low recent intake or potentially increased metabolic demand or retention by the body.
While boron is not officially classified as an “essential” nutrient for humans in the same way as iron or calcium, a growing body of evidence suggests it plays important roles in bone health, hormone balance, brain function, and inflammation regulation. Low levels can therefore carry meaningful implications for overall wellness.
Potential causes of low boron
-
Inadequate dietary intake
-
Most people get boron through plant-based foods like fruits (apples, grapes, pears), vegetables (especially leafy greens), legumes, and nuts. A diet low in these foods—such as a heavily processed or animal-based diet—can result in lower boron levels.
-
-
Poor absorption
-
Digestive disorders such as celiac disease, inflammatory bowel disease (IBD), or gut dysbiosis may interfere with the absorption of minerals, including boron.
-
-
Increased physiological need
-
During periods of inflammation, hormonal shifts (such as menopause), or bone remodeling, the body may use more boron, potentially lowering circulating or excreted levels.
-
-
Lack of supplementation
-
People who do not supplement and have limited boron-rich foods in their diet may show lower urinary excretion simply due to low overall exposure.
-
-
Chronic stress or toxin exposure
-
Ongoing oxidative stress or toxic burden may increase the body’s need for boron as it participates in antioxidant and detoxification pathways.
-
Why low boron might matter
Bone and joint health
Low boron may reduce the body’s ability to metabolize and retain calcium, magnesium, and vitamin D—three nutrients essential for bone density and skeletal health. Long-term deficiency has been associated with a higher risk of bone loss, osteopenia, or osteoporosis, especially in postmenopausal women or aging adults.
Hormonal imbalances
Boron plays a regulatory role in the balance of sex hormones, particularly estrogen and testosterone. Low boron status has been linked to reduced free testosterone in men and impaired estrogen balance in women, potentially contributing to symptoms such as fatigue, mood swings, or decreased libido.
Cognitive function
Several studies have shown that low boron intake can negatively affect brain function, leading to poorer short-term memory, reduced coordination, and diminished alertness. These symptoms are subtle but may be especially relevant in individuals dealing with brain fog, fatigue, or neurological concerns.
Inflammation and immune stress
Boron may help regulate inflammation by influencing enzymes like COX and modulating the activity of inflammatory cytokines. A deficiency may contribute to chronic low-grade inflammation, joint discomfort, or impaired immune responses.
Symptoms potentially associated with low boron
While boron deficiency is not often diagnosed clinically, some signs and symptoms may align with low boron status:
-
Joint stiffness or discomfort
-
Brittle bones or early signs of bone loss
-
Hormonal symptoms (e.g., PMS, low libido, fatigue)
-
Cognitive sluggishness or brain fog
-
Poor wound healing
-
Muscle cramps (especially when other minerals like magnesium are also low)
What to do if your boron level is low
-
Review your diet
Increase intake of boron-rich foods such as:-
Apples, pears, grapes, prunes, raisins
-
Avocados
-
Almonds, walnuts, and other nuts
-
Lentils and legumes
-
Leafy greens (spinach, kale)
-
Wine (in moderation)
-
-
Consider supplementation
Low-dose boron supplements (typically 1–3 mg/day) may be helpful under the guidance of a healthcare provider, especially in individuals at risk for bone loss or hormonal imbalance. -
Address gut health
If poor absorption is suspected, consider evaluating and supporting digestive function through testing and interventions targeting the gut microbiome and nutrient uptake. -
Balance other minerals
Since boron interacts with calcium, magnesium, and vitamin D, it’s important to assess these nutrients as well—either through laboratory testing or clinical history.
Summary
Low urinary boron levels on the Essential Elements panel may reflect insufficient intake, poor absorption, or increased physiological demand. Though not yet considered an essential nutrient in official guidelines, boron supports key systems including bone health, hormonal balance, cognitive function, and inflammation regulation. If your level is low, your healthcare provider may recommend dietary improvements, targeted supplementation, or further investigation into your nutrient absorption and metabolic health.
Article Review & Sources
All our content is backed by peer-reviewed studies, academic research, and trusted medical sources. We're committed to accuracy and transparency — see our editorial policy for details.
Laboratories
Bring All Your Lab Results Together — In One Place
We accept reports from any lab, so you can easily collect and organize all your health information in one secure spot.
Pricing Table
Gather Your Lab History — and Finally Make Sense of It
Finally, Your Lab Results Organized and Clear
Personal plans
$79/ year
Advanced Plan
Access your lab reports, explanations, and tracking tools.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 5 reports entered for you
- Cancel or upgrade anytime
$250/ once
Unlimited Account
Pay once, access everything—no monthly fees, no limits.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 10 reports entered for you
- No subscriptions. No extra fees.
$45/ month
Pro Monthly
Designed for professionals managing their clients' lab reports
- Import lab results from any provider
- Track lab results for multiple clients
- Customize reference ranges per client
- Export lab histories and reports
- Begin with first report entered by us
- Cancel or upgrade anytime
About membership
What's included in a Healthmatters membership
 Import Lab Results from Any Source
Import Lab Results from Any Source
 See Your Health Timeline
See Your Health Timeline
 Understand What Your Results Mean
Understand What Your Results Mean
 Visualize Your Results
Visualize Your Results
 Data Entry Service for Your Reports
Data Entry Service for Your Reports
 Securely Share With Anyone You Trust
Securely Share With Anyone You Trust
Let Your Lab Results Tell the Full Story
Once your results are in one place, see the bigger picture — track trends over time, compare data side by side, export your full history, and share securely with anyone you trust.





Bring all your results together to compare, track progress, export your history, and share securely.
What Healthmatters Members Are Saying
We implement proven measures to keep your data safe.
At HealthMatters, we're committed to maintaining the security and confidentiality of your personal information. We've put industry-leading security standards in place to help protect against the loss, misuse, or alteration of the information under our control. We use procedural, physical, and electronic security methods designed to prevent unauthorized people from getting access to this information. Our internal code of conduct adds additional privacy protection. All data is backed up multiple times a day and encrypted using SSL certificates. See our Privacy Policy for more details.

















