Lab Results Explained and Tracked
What does it mean if your Cesium result is too high?
Very high levels of Cs in urine are often associated with the use of cesium chloride as a questionable anti-cancer treatment. Cesium is a naturally- occurring element found in rocks, soil and dust at low concentrations. It is present in the environment only in the stable form of Cs133; the radioactive isotopes 134Cs and 137Cs are not measured or reported by Doctor's Data. Natural deposits of Cs ores occur in Main, South Dakota and Manitoba (Bernic Lake), Canada. Cesium may bio-accumulate in aquatic food chains; higher levels of cesium have been found in Pacific deep-sea fish and local shellfish since the 2011 Fukoshima reactor accident. Cesium may be used in high-density drilling fluids (oil and gas industry) and may contaminate local water and vegetation; Cs has been found in cow's milk. Cesium may occur naturally in mineral waters; one study analyzed the Cs concentration in 163 mineral and thermal waters and found the level ranged from 4.5 to 148 μg per liter.
Cesium can be absorbed after oral ingestion, upon breathing contaminated air and through contact with the skin. Cesium is readily absorbed across the brush border of the intestines in a manner similar to potassium and most is eventually excreted through the urine and feces. The biological half-life of Cs in humans ranges from 15 days in infants to 100-150 days in adults.
The cesium-137 isotope is used in cancer treatments, for ventricular function and pulmonary imaging studies, industrial radiology, and for food and instrument sterilization; Cs137 agents may contain small amounts of Cs133. Non-radioactive cesium chloride may be advertised on the internet as "high pH therapy." Currently there is no support in the scientific literature for that purpose as advertised. Radioactive Cs isotopes may contaminate soil at nuclear waste sites. Cesium may be used in industry for the production of photoelectric cells, vacuum tubes, spectrographic instruments, scintillation counters, DNA biochemistry, in various optical or detecting devices.
Target organs of potential toxic effects of Cs are the liver, intestine, heart, and kidneys. Physiological effects of excessive Cs include ventricular arrhythmias and displacement of potassium from muscle cells and erythrocytes. Cesium can have significant effects on both the central and peripheral nervous systems. Cesium may cause epileptic seizures because it can share the same receptor as the excitatory bioamine glycine. Cesium can interfere with active ion transport by blocking potassium channels and also can interfere with lipid metabolism. Excessive Cs may modify plasma membrane integrity, alter cytoplasmic components and cause cytogenetic damage.
It is unlikely that children or adults would be exposed to enough Cs133 to experience any health effects that could be related to the stable Cs itself. Animals given very large doses of Cs compounds have shown changes in behavior, such as increased activity or decreased activity, but it is unlikely that a human would be exposed to enough stable Cs to cause similar effects.
The isotope Cs137 is used in radiation therapy for certain types of cancer. Other medical uses of Cs are monitoring left ventricular function with Cs137 iodide probes and monitoring pulmonary endothelial permeability with Cs137 iodide crystal mini-detectors. Again, it is emphasized that Cs measured at Doctor's Data is Cs133, not Cs137. Environmental contamination by Cs137 as a result of radioactive fallout could be a concern. Exposure to Cs may be assessed by hair elemental analysis.
Commonly used chelating agents are not effective binders of Cs.
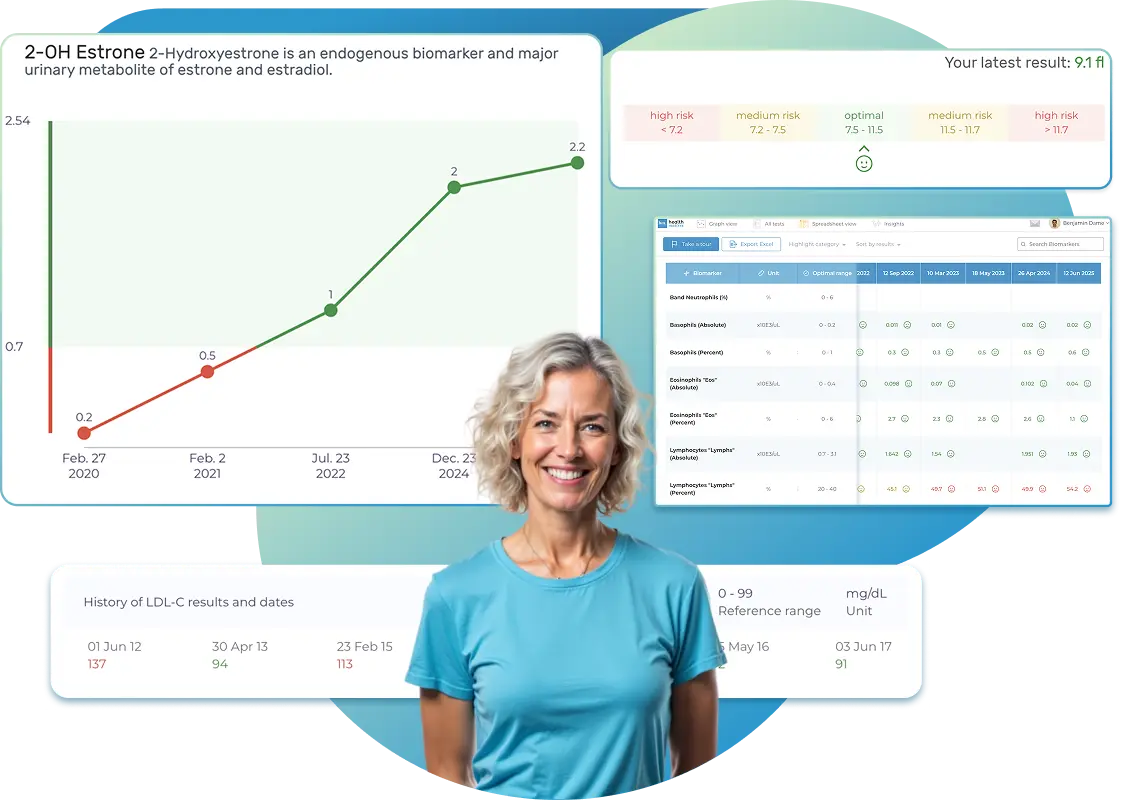
All Your Lab Results.
One Simple Dashboard.
Import, Track, and Share Your Lab Results Easily
Import, Track, and Share Your Lab Results
Import lab results from multiple providers, track changes over time, customize your reference ranges, and get clear explanations for each result. Everything is stored securely, exportable in one organized file, and shareable with your doctor—or anyone you choose.
Cancel or upgrade anytime
Article Review & Sources
All our content is backed by peer-reviewed studies, academic research, and trusted medical sources. We're committed to accuracy and transparency — see our editorial policy for details.
Laboratories
Bring All Your Lab Results Together — In One Place
We accept reports from any lab, so you can easily collect and organize all your health information in one secure spot.
Pricing Table
Gather Your Lab History — and Finally Make Sense of It
Finally, Your Lab Results Organized and Clear
Personal plans
$79/ year
Advanced Plan
Access your lab reports, explanations, and tracking tools.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 5 reports entered for you
- Cancel or upgrade anytime
$250/ once
Unlimited Account
Pay once, access everything—no monthly fees, no limits.
- Import lab results from any provider
- Track all results with visual tools
- Customize your reference ranges
- Export your full lab history anytime
- Share results securely with anyone
- Receive 10 reports entered for you
- No subscriptions. No extra fees.
$45/ month
Pro Monthly
Designed for professionals managing their clients' lab reports
- Import lab results from any provider
- Track lab results for multiple clients
- Customize reference ranges per client
- Export lab histories and reports
- Begin with first report entered by us
- Cancel or upgrade anytime
About membership
What's included in a Healthmatters membership
 Import Lab Results from Any Source
Import Lab Results from Any Source
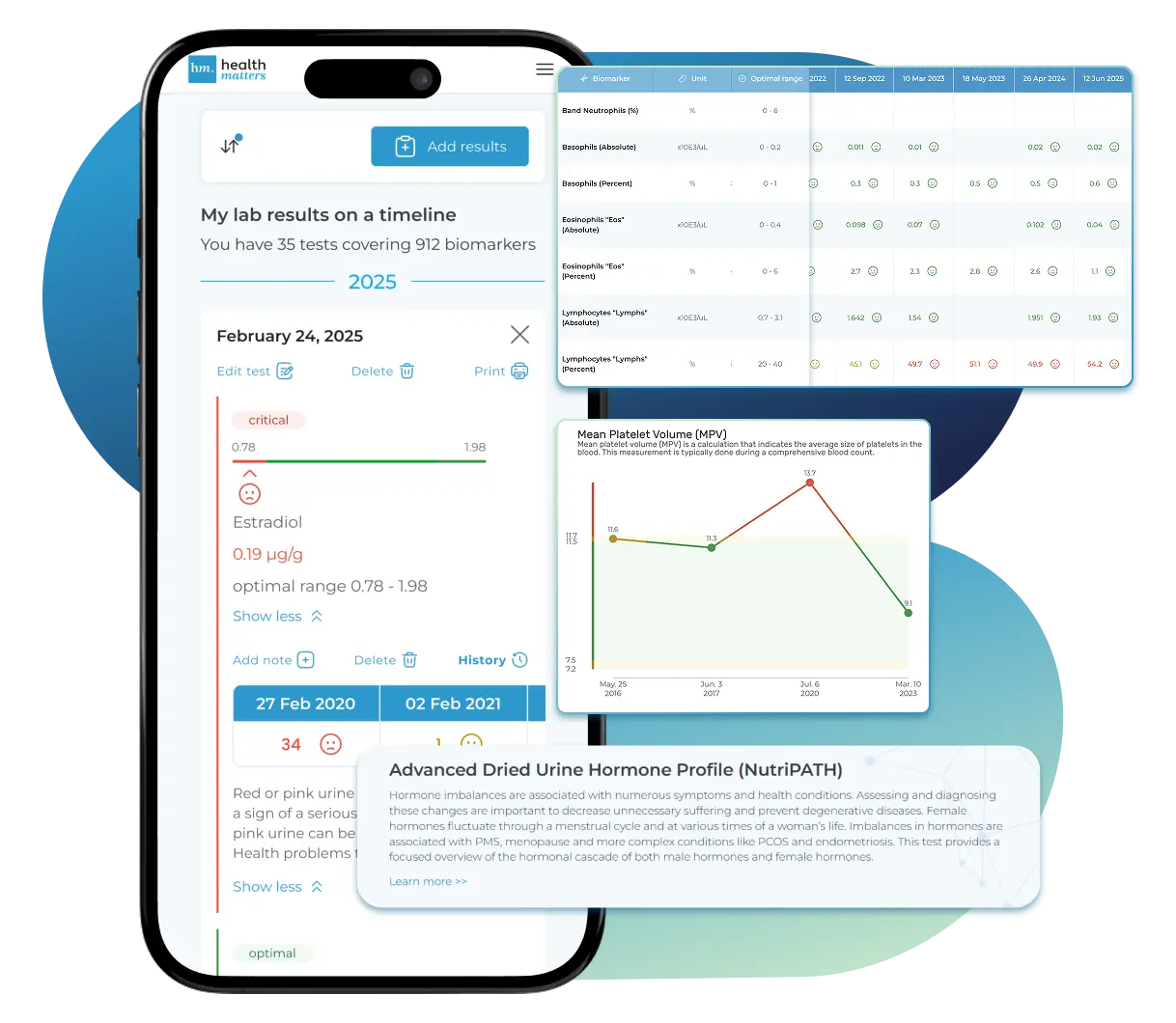
 See Your Health Timeline
See Your Health Timeline
 Understand What Your Results Mean
Understand What Your Results Mean
 Visualize Your Results
Visualize Your Results
 Data Entry Service for Your Reports
Data Entry Service for Your Reports
 Securely Share With Anyone You Trust
Securely Share With Anyone You Trust
Let Your Lab Results Tell the Full Story
Once your results are in one place, see the bigger picture — track trends over time, compare data side by side, export your full history, and share securely with anyone you trust.





Bring all your results together to compare, track progress, export your history, and share securely.
What Healthmatters Members Are Saying
We implement proven measures to keep your data safe.
At HealthMatters, we're committed to maintaining the security and confidentiality of your personal information. We've put industry-leading security standards in place to help protect against the loss, misuse, or alteration of the information under our control. We use procedural, physical, and electronic security methods designed to prevent unauthorized people from getting access to this information. Our internal code of conduct adds additional privacy protection. All data is backed up multiple times a day and encrypted using SSL certificates. See our Privacy Policy for more details.

















